Research Highlights
No Relaxing in the Fight Against Liver Cancer
Institutions:
Lanzhou University, China
Team:
Wei, J., Yao, J., Yang, C., Mao, Y., Zhu, D., Xie, Y., Liu, P., Yan, M., Ren, L., Lin, Y., Zheng, Q., & Li., X.
Disease Model:
Liver Cancer
Hydrogel:
VitroGel® 3D
Cancer cell stiffness was measured by atomic force microscopy in tumors taken from liver cancer patients. The findings suggest that there are additional factors that must be analyzed when predicting whether cells will turn cancerous.
Increased cell stiffness, or the ability of cells to resist elastic deformation under force, can be a characteristic of cancer cells. A stiffer structure arises when the extracellular matrix (ECM) loses its ability to be controlled by usual genetic factors, and an overabundance of protein cross-linking takes place. Such a scenario is reflective of what is known as the “tumor microenvironment” or TME. Importantly, the TME can determine whether cells, including liver cells, return to a stem cell state, and develop into tumors. There is thus a relationship between cell stiffness and the likelihood that a tumor will arise in a tissue, but the details of this relationship are not clear.
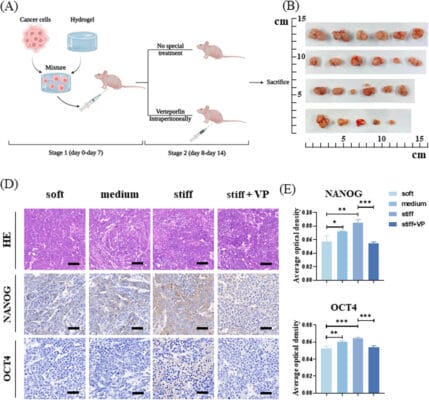
A team of oncologists from Lanzhou University in China set out to find a means of accurately quantifying cell stiffness, and then determine its genetic and environmental determinants. It was believed that a specific protein, the yes-assisted protein (YAP) may be an important component in determining the state of the ECM that would cause it to stiffen and was therefore an addition target of this team’s studies. The authors decided to use atomic force microscopy (AFM) to assay stiffness, measured in Pascals (Pa), in hepatocellular carcinoma (HCC) cells extracted from three hospital patients. AFM is a very sensitive technique that measures the vibration of a tiny stylus as it moves over a surface, akin to a microscopic record player. Because stiffness was expected to vary across a tissue, the authors first measured the stiffnesses of three randomly-chosen regions on a liver cancer tumor and found that they were indeed non-uniform; the three sites registered stiffness values of about 1000 Pa, 4500 Pa, and 9300 Pa. These differences scaled roughly with the amount of collagen in the sample and with the degree of protein crosslinking.
Next, to simulate TMEs with these stiffness values, the authors used TheWell Bioscience’s VitroGel® 3D, mixed at different dilution ratios with the provided Dilution Solution, and then coated with polyacrylamide, to create hydrogels with three stiffness ranges: 12,000 Pa (hard, i.e., stiff), 3600–6000 Pa (medium), and 600–1500 Pa (soft). They then grew HCCs in these three media and found that cells grown on medium and stiff hydrogel had greater surface areas and more apparent networks of actin filament networks growing out of them. Thus, they could conclude that cell growth characteristics and the stiffness of the TME were definitely related.
Lastly, the authors used RT-PCR to measure YAP-related gene expression of the cells that possessed the different stiffnesses. While YAP mRNA expression levels per se did not vary, there was a clear increase in the levels of two downstream gene products related to YAP (CTGF and ANKRD proteins), indeed implicating YAP in determining cell stiffness.
The data presented in this study demonstrated that it was feasible to measure cancer cell stiffness, and that there were genetic and phenotypic correlates with this parameter. The authors uncovered a potential mecahotranscution pathway whereby cellular changes lead to an effect on the local TME, which then could abet tumor growth. These findings are valuable, but they point to an increased layer of complexity affecting tumorigenesis and cancer heterogeneity that mandates further study.


